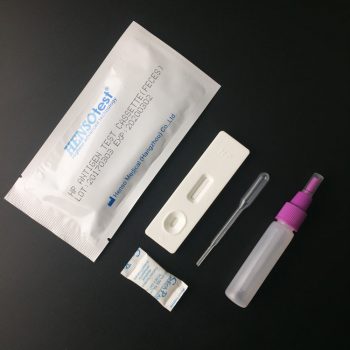
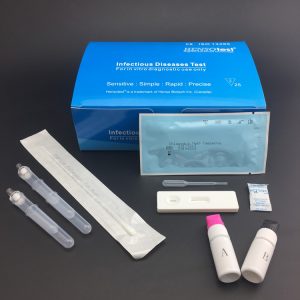
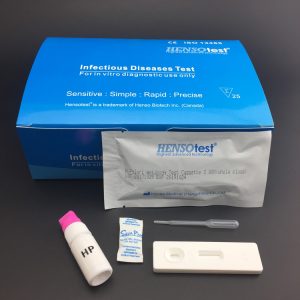
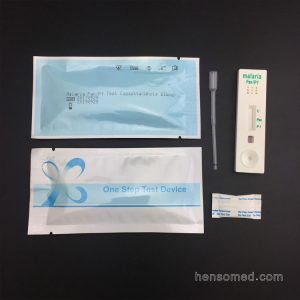
H.pylori Antigen Stool Test Cassette
- Format: Cassette
- Size: 3.0mm, 4.0mm
- Specimen: Feces
- Accuracy over 99%
- Results read in 15 minutes
- Methodology: Colloidal gold
- Shelf time: 2 years
- Description
- Instruction for Use
- Ordering Information
- Inquiry
H.pylori Antigen Test Cassette detection in stool is a rapid, non invasive, easy to perform test that can be used to detect active infection, monitor effectiveness during therapy and to confirm cure after antibiotic use. The ease to collect the specimen, especially in children, in whom endoscopy would be difficult and the non requirement of specially trained staff to collect and perform the test adds to the tests advantage. Also, prior preparation of the patient is not necessary unlike in upper gastrointestinal endoscopy. The sensitivity and specificity of this test are better than the other detection methods
Features
- Quick result read in 15 minutes
- High accuracy over 99%
- Sensitivity 98.3%
Packaging
Box of 20T, Ctn of 800T
Helicobacter Pylori Antigen (HP Ag)
Rapid Test Cassette
【SUMMARY AND EXPLANATION】
H.pylori antigen detection in stool is a rapid, non invasive, easy to perform test that can be used to detect active infection, monitor effectiveness during therapy and to confirm cure after antibiotic use. The ease to collect the specimen, especially in children, in whom endoscopy would be difficult and the non requirement of specially trained staff to collect and perform the test adds to the tests advantage. Also, prior preparation of the patient is not necessary unlike in upper gastrointestinal endoscopy. The sensitivity and specificity of this test are better than the other detection methods
【PRINCIPLE OF THE TEST】
This assay is a double antibodies chromatographic lateral flow immunoassay. The test strip in the device includes: 1) a burgundy-colored conjugate pad containing colloidal gold coupled with H. pylori ( out of membrane protein, OMP) polyclonal antibodies, and Nitrocellulose membrane containing a test line ( T - line) and a control line (C- line). The T line is coated with H. pylori antibody, and the C- line is coated with goat anti-H. pylori antibody. The antigens used in this device are from H. pylori cell lysate. When patient antigen to H. pylori are present in the specimen, the T line will become a burgundy-colored band . If antigen to H. pylori are not present or are present below the detectable level, no T line will develop. The C line should always appear as a burgundy-colored band regardless of the presence of antigen to H. pylori. The C line serves as an internal qualitative control of the test system to indicate that an adequate volume of specimen has been applied and the flow occurred.
【SPECIMEN COLLECTION】
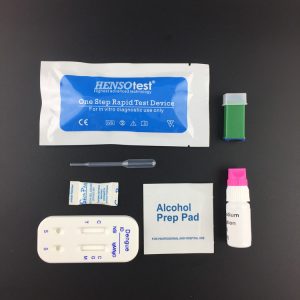
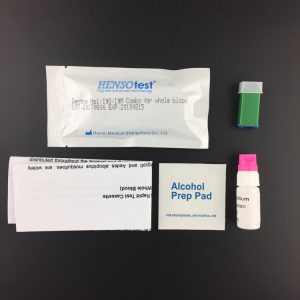
- Collect stool sample by using the special sample collection device provided. Firstly,take out the sample collection stick, and collect the sample by dipping the stick into 3-6 different places (around 5mm of stool spot) of the stool sample.Then, put the sample collection stick into the tube(the sample collection device) and shake it several times.
- Attention: The sample may be stored at 2-8°C for 48 hours, at -20°C for long term storage. Bring the sample back to the room temperature before testing.
【TEST PROCEDURE】
- Remove the test device from its foil pouch by tearing along the notch.
- Specimen collection: Dispense 0.5 ml of working sample buffer into a sample tube. Please see to Specimen collection chart. Shake the sample collection tube upright; Mix for 20 seconds.
- Using the plastic pipettor provided, draw 2 or 3 drops of sample into the sample well of the cassette. as in the illustration.
- Read the test results within15minutes (Some positive results may be seen earlier)
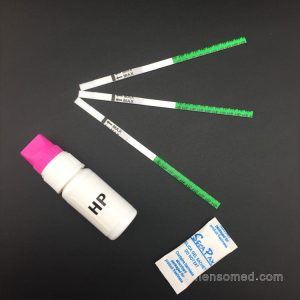
【INTERPRETATION】
- Positive: In addition to a pink colored Control band, a distinct pink colored band also appears on the Test region.
- Negative:Only one colored band appears on the Control region. No apparent band on the Test region.
- Invalid: A total absence of color on both regions is an indication of procedure error and/or test reagent deterioration.
【WARNINGS AND PRECAUTIONS】
- For In Vitro Diagnostic Use only.
- Follow clinical and/or laboratory safety guidelines in collection, handling, storage, and disposal of patient specimens, and all items exposed to patient specimens.
- The Sample Buffer Solutions contain a saline solution with a bacteriocide (sodium azide) and a detergent at low concentrations. If the solution comes in contact with the skin or eyes, flush with ample volumes of water.
- Solutions that contain sodium azide may react with lead or copper plumbing to form potentially explosive metal azide. Large quantities of water must be used to flush discarded solutions down a sink.
- Store sealed HP Ag test kit at room temperature, in a dry place away from direct sunlight. Avoid excessive heat (>30°C).
- Do not freeze. Don’t use tests kit after expiration date.
| REF. | Description | CBM/ctn | KGS/ctn |
| L115402 | HP antigen test cassette 3.0mm for feces | 0.069 | 10 |
| L115403 | HP antigen test cassette 4.0mm for feces | 0.069 | 10 |