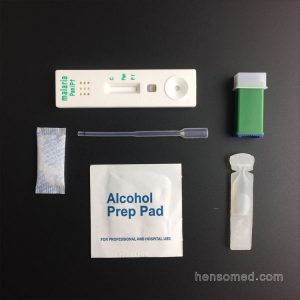
Malaria P.f./P.v. Test Cassette
- Format: Cassette
- Size: 3.0mm, 4.0mm
- Specimen: Whole Blood/Serum/Plasma
- Accuracy over 99.9%
- Results read in 20 minutes
- Methodology: Colloidal gold
- Shelf time: 2 years
- Description
- Instruction for Use
- Ordering Information
- Inquiry
HENSOtest Malaria P.f./P.v. Test Cassette is a rapid chromatographic immunoassay for the qualitative detection of two kinds of circulating plasmodium falciparum (P. falciparum (P.f.) and P. vivax (P.v.) in whole blood.
Malaria is a serious parasitic disease characterized by fever, chills, and anemia and is caused by a parasite that is transmitted from one human to another by the bite of infected Anopheles mosquitoes. There are four kinds of malaria that can infect humans: Plasmodium falciparum, P.vivax, P.ovale and P.malariae. In humans, the parasites migrate to the liver where they mature and release another form, the merozoites.
Features
- Result read in 20 minutes
- High accuracy 99.9%
- Sensitivity 99.9%
- Specificity 99.9%
Packaging
Box of 25T, Ctn of 1000T
One Step Malaria pf(HRPⅡ)/Pv/PF(pLDH)Antigen Detection Test Device (Whole Blood)
Intended use:
For the rapid qualitative determination of Malaria P.falciparum specific histidine rich protein-2 (Pf HRP-2) and malaria P.vivax specific lactate dehydrogenase (pLDH) in human blood as an aid in the diagnosis of Malaria infection.
Summary
Malaria is a serious parasitic disease characterized by fever, chills, and anemia and is caused by a parasite that is transmitted from one human to another by the bite of infected Anopheles mosquitoes. There are four kinds of malaria that can infect humans: Plasmodium falciparum, P.vivax, P.ovale, and P.malariae. In humans, the parasites migrate to the liver where they mature and release another form, the merozoites.
The One-Step Malaria pf(HRP II)/Pv/PF(pLDH)Antigen Detection Test Device(Whole Blood) contains a membrane strip,which is pre-coated with two monoclonal antibodies as two separate lines across a test strip.One monoclonal antibody(test line 1)is specific to the p.falciparum histidine rich protein-2(Pf HRP-2) and another monoclonal antibody(test line 2) is pan specific to the lactate dehydrogenase of Plasmodium species(P.falciparum,vivax,malariae,ovale).Conjugate pad is dispensed with monoclonal antibodies conjugated to colloidal gold,which are specific to P.falcparum histidine rich protein-2(Pf HRP-2)and pan specific to the lactate dehydrogenase of Plasmodium species.
Materials:
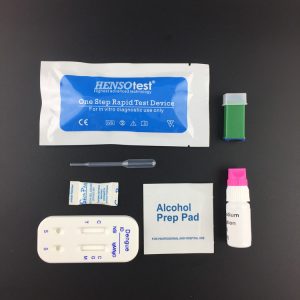
Materials Provided
Test Device
Assay Buffer
Instructions for Use
5ul sample loop
Materials not Provided
Calibrated pipette
Lancet
Timer
Specimen Collection
Collect whole blood into a collection tube (containing EDTA, citrate or heparin) by venipuncture.
If specimens are not immediately tested, they should be refrigerated at 2~8°C. For storage periods greater than three days, freezing is recommended. They should be brought to room temperature prior to use. Using the specimen after long-term storage of more than three days can cause non-specific reaction.
When stored at 2~8°C, the whole blood sample should be used within three days.
Directions for Use:
- Use sample loop or pipette to add 5μl of whole blood into sample well(A).
- Add 2 drops (60μl) of assay buffer into well(B).
- Read the test result within 20 minutes.
Interpretation of Results:
Positive:
P.falciparum malaria
In addition to a pink / purple colored control (C) band, a pink / purple colored band appear at the (P.f) region respectively. The result is P.f positive.
P.vivax malaria:
In additional to a pink / purple colored control(C) band, one pink / purple band appears at the (P.v) region.
P.vivax malaria and P. falciparum malaria
Negative: Only one pink/purple band appears on the control (C) region. No apparent band on the test region (Pv/Pf)
Invalid: A total absence of color in both regions is an indication of procedure error and/or the test reagent has been deteriorated.
Precautions:
- For professional in vitro diagnostic use only, Do not use after expiration date.
- Do not eat, drink or smoke in the area where the specimens or kits are handled.
- Handle all specimens as if they contain infectious agents. Observe established precoutions against mocrobiological hazards throughout testing and follow the standard procedures for proper disposal of specimens.
- Wear protective clothing such as laboratory coats, disposable gloves and eye protection when specimens are being tested.
- Humidity and temperature can adversely affect results.
Storage and Stability:
The kit can be stored at room temperature or refrigerated(2-30°C)。
The test device is stable through the expiration date printed on the sealed pouch.
The test device must remain in the sealed pouch until use. Do not freeze. Do not use beyond the expiration date.
| REF. | Description | CBM/ctn | KGS/ctn |
| L114601 | Malaria Pf/Pv Test cassette 3.0mm for whole blood/serum/plasma | 0.069 | 9 |
| L114602 | Malaria Pf/Pv Test cassette 4.0mm for whole blood/serum/plasma | 0.069 | 9 |