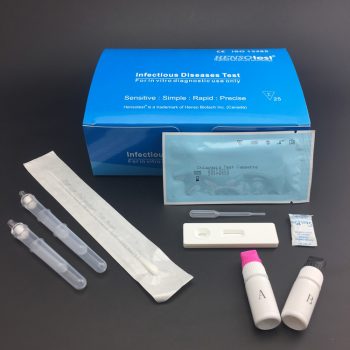
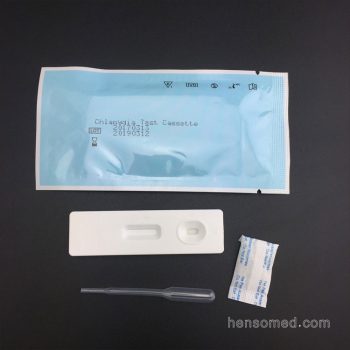
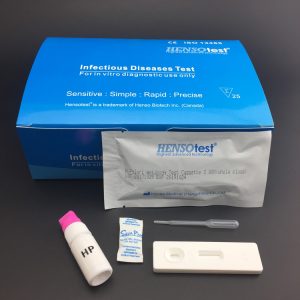
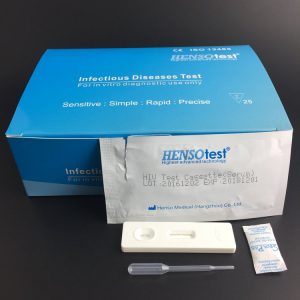
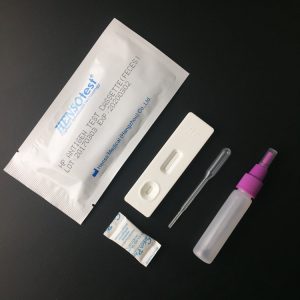
Chlamydia Screen Rapid Test Kit
- Format: Cassette
- Specimen: Cervical Mucus
- Size: 3.0mm, 4.0mm
- Accuracy over 98.2%
- Results read in 10 minutes
- Methodology: Colloidal gold
- Shelf time: 2 years
- Description
- Instruction for Use
- Ordering Information
- Inquiry
Chlamydia Test Kit is a rapid visual immunoassay for the qualitative presumptive detection of Chlamydia trachomatis in female cervical, male urethral and male urine specimens. This kit is intended for use as an aid in the diagnosis of Chlamydia infection.
Complete Kit contain:
Test Cassette + Dropper + Desiccant + Buffer + Cervical Swab + Reagent A & Regent B
Features
- Quick result read in 10 minutes
- High accuracy over 98.2%
- Complete kit with accessories for easy use at home
Operating instruction for Diagnostic kit for Chlamydia (Colloidal Gold)
【Product Name】
The common name:Chlamydia Rapid Test Kit
The English name: Diagnostic kit for Chlamydia (Colloidal Gold)
【Packing Model Specification】Cassette Type
【Intended use】
This product is used for qualitative detection of the presence of chlamydia in female cervix and male urethra, and the clinical auxiliary diagnosis of chlamydia infection, and test results also need to be further confirmed by the clinician in connection with symptoms, signs and other test results.
【Detection Principle】
Chlamydia diagnostic kit uses highly specific antigen-antibody reaction and immune chromatography analysis techniques for the qualitative detection of whether clinical specimens contain chlamydia trachomatis antigen, and the kit contains chlamydia trachomatis antigen-specific antibodies, which are pre-fixed on the test area (T) on the membrane, and the corresponding antibodies on the quality control area (C). During the detection, the pyrolysis-off specimens on the cotton swab are titrated into the sampling wells (S) of the kit, and the specimens make a specific antibody reaction with the chlamydia trachomatis antigen of the pre-coated gold particles. Then, the mixture followed up with upward chromatography under capillary effect. If it is positive, gold-labeled Chlamydia will first bind with Chlamydia trachomatis antigen in the specimens in the chromatography process, then the bonder will be combined by specific antibody of Chlamydia trachomatis antigen, which is fixed on the membrane, and within the test area (T) there will appear a red band. If it is negative, there will no red band within the test area (T). Regardless of whether the Chlamydia trachomatis antigen exists in clinical samples, there will be a red band within the quality control area (C). The red band within the quality control area (C) is to determine whether there is sufficient sample, and whether the process of chromatography is in line with the normal standards, and at the same time it is used as the internal control standards of the agent.
【Major component ingredients】
- Chlamydia diagnostic kit
- A copy of operating instruction
- Lysate A
- Lysate B
【Sample collection】
The quality of the collected specimens is of paramount importance for chlamydia detection. The detection quality of chlamydia depends on the accurate sample collection techniques, which should make samples contain a large number of a certain active cellular components, and the samples should not just contain body fluids.
Female cervical sampling:
- Use sterile cervical swabs, or sterile linen, polyester swabs. Before sampling, wipe mucus of the cervical extraoral region with other swabs or cotton balls, and insert the sampling swab into the cervical tube through the squamous columnar epithelium junction, until the swab head has been virtually invisible. After rotating swab for 15-20 seconds, take it out, and do not hit the outside of the cervix and vaginal wall. This will ensure to get more columnar epithelial cells, while the chlamydia mainly parasite in columnar epithelial cells.
- Cervical samples can also be collected by the use of cell brush (not provided) (Note: Pregnant women should not use this method). After cleaning the cervical extraoral region, insert a cell brush into the cervical tube, through the scale columnar epithelial cells junction, and stay for 2-3 seconds, and after two cycles of rotating brush cells, take it out, and be careful not to hit the vaginal wall.
- If the detection can be immediately carried out, please place the swab in the samples processing tube after sampling.
Male urethral specimen collection:
Urethral-use swabs or cells brush (not provided) can be used for urethral sampling. Patients should not urinate within at least 1 hour before sampling.
Insert the swab or cell brush into the urethra for 2-4 cm, and after rotating for 3-5 seconds, take it out. If the detection can be immediately carried out, please place the swab in the samples processing tube after sampling. If you don’t take an immediate test, place the sample in dry test tube for storage or transportation.
Sample can be kept for 4-6 hours at room temperature (15-30 ℃), or (2-8 ℃) kept under cold-storage for 24-72 hours. It can not be frozen. All specimens should revert to room temperature before detection. We propose that the experiment should be made immediately after sample collection.
【Test procedure】
- Samples processing and control:
processing of cervical swabs and urethral swabs:
- Place the sample processing tubes on the worktable, and add 6 drops of solution A.
- Put the sampling swab into the sample processing tube, which contains solution A, and store at room temperature, and continuously rotate and squeeze the swab in the tube wall in the process, so that the liquid is constantly being squeezed out, and repeat it for several times and deal with it for 2 minutes.
- Then add 6 drops of solution B, and rotate and squeeze swab, as far as possible make the liquid flow out, and then discard the swab in line with the handling method of infected goods.
- If the samples shall be used, within 60 minutes after treated, this will not affect detection results of the kit.
Notes: The dropping amount of solution A and solution B should be equal, when the swabs is processed
B. Detection steps:
- Please read the instructions for the test kit carefully before operation.
- Take out the test kit from the sealed bag, and place it in a clean, dry and level worktable, and label the sample number or name. If the test kit is stored at the place, which temperature is lower than room temperature, and the test kits and reagents required to be taken out in advance, and it should revert to room temperature before it is being used.
- Draw 2 or 3 drops of processed samples in the sample processing tube should be dripped into the sampling wells of the test kit.
- Wait for the appearance of the results. 10 minutes after the sample is dripped, the results can be interpreted. The time of the red line varies in line with the contents of chlamydia, which is collected with swabs, and some positive samples may show the result after 60 seconds. In order to ensure the negative results, please do not judge the result after 15 minutes.
【Results determination】
Positive: Two purple bands appear at the test line and the quality-control control lines of the test paper card. The positive results show that: samples contain chlamydia trachomatis antigen.
Negative: only one purple band appears at the quality-control control line of the test paper card. Negative results show that: Chlamydia trachomatis antigen can’t be detected in samples, or the content is below the detectable range. Patient specimens should be confirmed no Chlamydia trachomatis infection by culture. If the culture results and test results are inconsistent, and take another sample to culture.
【The interpretation of test results】Invalidity: If there is no purple bands at the quality-control control line, it indicates that the operation course is incorrect or the kit fails. In this case, please re-read the instructions carefully and use the new test paper cards for re-test. If there are still problems, the batch product should be immediately stopped use, and please contact with local suppliers.
- The purple bands, which appear at the quality-control control lines, is used as the internal control standards of the test paper strip.
- The purple bands appear gradation phenomenon at the testing line, when testing different specimens, and this is due to high and low concentrations of surface antigen. However, within the specified time, regardless of the depth of the ribbon color, even if only a very weak ribbon should also be judged as positive results.
【The limitations of the testing methods】
- The detection of Chlamydia trachomatis with this kit depends on the content of chlamydia in the sample, sampling methods and the patient’s condition: such as age, venereal disease history, whether there is asymptom and other conditions shall affect the detection.
- If determining the chlamydia of different serotypes with this kit, the minimum detectable level shall be also different.
- May be due to operational improper of technology or steps, and the presence of other non-listed drugs, which may interfere with detection, and it shall interfere with detection and lead to inconsistent or erroneous results. Please refer to the column
【Performance indicators of products】
- Minimum detectable quantity
If taking quality control test with the E-type chlamydia, the minimum detectable quantity should be no higher than 4 × 103 IFU / ml. - Specificity
The following micro-organisms do not cross-react with the chlamydia test kit when they are
1 × 106CFU/ml:
The types of the micro-organisms and the concentration of the tested bacteria (CFU / ml)
ACINETOBACTER 106
SALMONELLA TYPHI 106
NEISSERIA GONORRHOEAE 106
Staphylococcus aureus 106
PSEUDOMONAS 106
CANDIDA ALBICANS 106
ESCHERICHIA COLI 106
STREPTOCOCCUS FAECALIS 106
STREPTOCOCCUS FAECIUM 106
STREPT B 106
TRICHOMONAS VAGINALIS 106
【Instructions and Notes】
- Only used by extrinsic diagnosis and professionals.
- Do not use after the expiry; Don’t mix and match different batches of reagents or test kits; Do not mix the lids of solutions A and B.
- The collection, processing, storage, discarding of the sample and the use of the reagent in the kit should take appropriate precautions. All the samples, reagents, and control substance should be handled as infectious substances.
- After the completion of the test, the sample shall be sterilized at 121 ℃ high temperature for 20 minutes, or use 0.5-1.0% sodium hypochlorite (or household bleach) to soak for 1 hour before discarded.
- Solution A contains sodium hydroxide, and solution B contains hydrochloric acid, if these two solutions are splashed on the skin or in eyes, we should rinse immediately with plenty of water.
- Can only collect samples with sterile Dacron swab or cell brush, and cotton swab sampling is not available.
- The region of dealing with sample and reagent prohibits eating, drinking and smoking; when collecting and testing samples, you should wear an experimental clothing and gloves.
- The final diagnosis of the patient can not simply rely on the results of this experiment, the comprehensive judgments must be made combined with clinical examination by a doctor.
【Storage conditions and valid period】
Preserve at 2-30 ℃ in sealed dark dry place, and the valid period is 18 months.
【References】
- Control practical handbook for bio-products and their quality standards for raw and auxiliary materials, Volume III, national audio-visual publishing house, Miao Yong, Zang Guangzhou editor in chief.
- The preparation instructions for extrinsic diagnostic reagents guideline; State Food Drug Administration Device [2007] No. 240.
| REF. | Description | Packaging | CBM/ctn | KGS/ctn |
| L115621 | Chlamydia test cassette 3.0mm for Cervical Mucus | 25T/box, 1000T/carton | 0.069 | 9 |
| L115622 | Chlamydia test cassette 4.0mm for Cervical Mucus | 25T/box, 1001T/carton | 0.069 | 9 |